 This advocates for the increased role of theoretical methods in analysis of stereoelectronic effects. While the general trends revealed in this work should be useful for the qualitative understanding of stereoelectronic effects, one should bear in mind that the magnitude of hyperconjugative effects is extremely sensitive to small variations in structure and in substitution. This effect can be relied upon in the design of molecular diodes with sigma bridges with unidirectional electron conductivity. For example, C-chalcogen bonds are excellent sigma acceptors at the carbon end but poor sigma acceptors at the chalcogen end. Stereoelectronic effects displayed by C-X bonds with X from second and third periods are highly anisotropic. As a result, the acceptor ability of sigma bonds can be significantly modified by substitution and is conformer dependent. A sigma bond is created when specific combinations of atomic. Example 4: Identifying What Is and What Is Not a Sigma () Bond. The combination of several effects of similar magnitude influences acceptor ability of sigma bonds in monosubstituted ethenes in a complex way. Sigma bonds can be made through the combination of a few different hybridized and unhybridized bonding orbitals, but the sideways overlap of two parallel p-type atomic orbitals always produces a single pi bond. This simple picture of acceptor ability of sigma bonds being controlled by electronegativity in periods and by sigma orbital energy in groups is changed in monosubstituted ethenes where the role of electronegativity of the substituent X becomes more important due to increased overlap between sigma orbitals. Enhancement of acceptor ability of C-X sigma bonds as one moves from left to right in periods parallels the increase in electronegativity of X, whereas augmentation of acceptor ability in groups is opposite to the changes in electronegativity of X and in the C-X bond polarization, following instead the decrease in the energy of sigma(C)(-)(X) orbitals when one moves from the top to the bottom within a group. The acceptor ability of the C-X sigma bonds in monosubstituted ethanes increases when going to the end of a period and down a group. 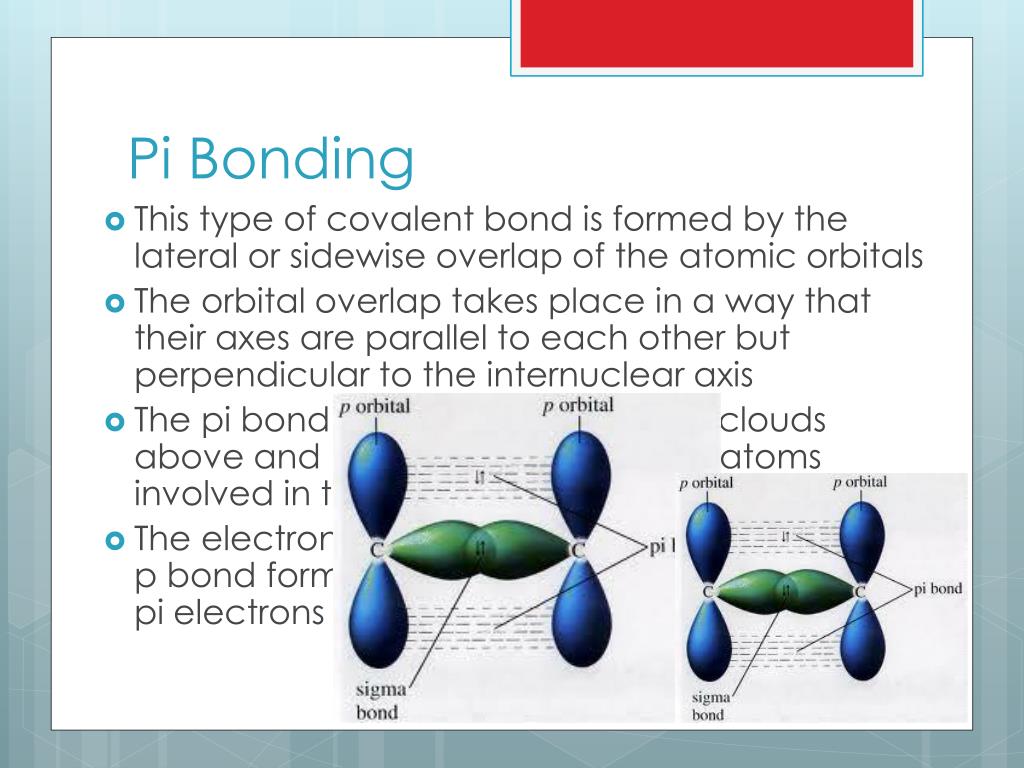
Combining the in-phase orbitals results in a bonding orbital.A systematic study of general trends in sigma acceptor properties of C-X bonds where X is a main group element from groups IVa-IIa is presented. One contains the axis, and one contains the perpendicular. Combining the out-of-phase orbitals results in an antibonding molecular orbital with two nodes. The reaction is illustrated by the exchange of. bond metathesis is a chemical reaction wherein a metal-ligand bond undergoes metathesis (exchange of parts) with the sigma bond in some other reagent. Side-by-side overlap of each two p orbitals results in the formation of two π molecular orbitals. Metal compounds with d 0 electron count are able to activate C-H bonds through bond metathesis reactions. For the out-of-phase combination, there are two nodal planes created, one along the internuclear axis and a perpendicular one between the nuclei.įigure 7.7.6. Electrons in this orbital interact with both nuclei and help hold the two atoms together, making it a bonding orbital. In molecular orbital theory, we describe the \pi orbital by this same shape, and a \pi bond exists when this orbital contains electrons. Definition and examples of pi bond The covalent bond formed by lateral overlap of atomic orbitals is called as pi bond. 
In valence bond theory, we describe π bonds as containing a nodal plane containing the internuclear axis and perpendicular to the lobes of the p–\pi orbitals, with electron density on either side of the node. The side-by-side overlap of two p orbitals gives rise to a pi (\pi) bonding molecular orbital and a \pi* antibonding molecular orbital, as shown in Figure 7.7.6. 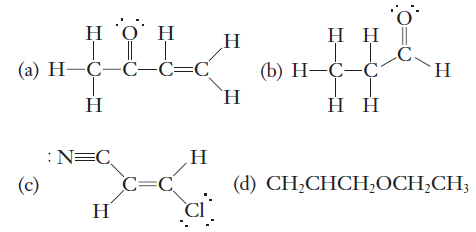
Combining wave functions of two p atomic orbitals along the internuclear axis creates two molecular orbitals, σp and σ∗p. Just as with s-orbital overlap, the asterisk indicates the orbital with a node between the nuclei, which is a higher-energy, antibonding orbital.įigure 7.7.5. There is an \ce^* (antibonding) (read as “sigma-p-x” and “sigma-p-x star,” respectively). This electronic structure adheres to all the rules governing Lewis theory. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
